‘Milestone’ at-home test for COVID and flu is approved. When can you buy it in Charlotte?
A first-of-its-kind at-home test that detects the viruses behind both COVID-19 and common strains of the flu has been approved for use in the U.S. and could soon be available to folks in Charlotte.
The company behind the new test hasn’t announced a U.S. release date for it yet, but the test is already available in other countries. And the same company already has other at-home COVID tests for sale in the U.S.
Its forthcoming release will be “a major milestone,” experts say.
Here’s what to know about the new test and when it will be available to Charlotte families:
How does the flu and COVID test work?
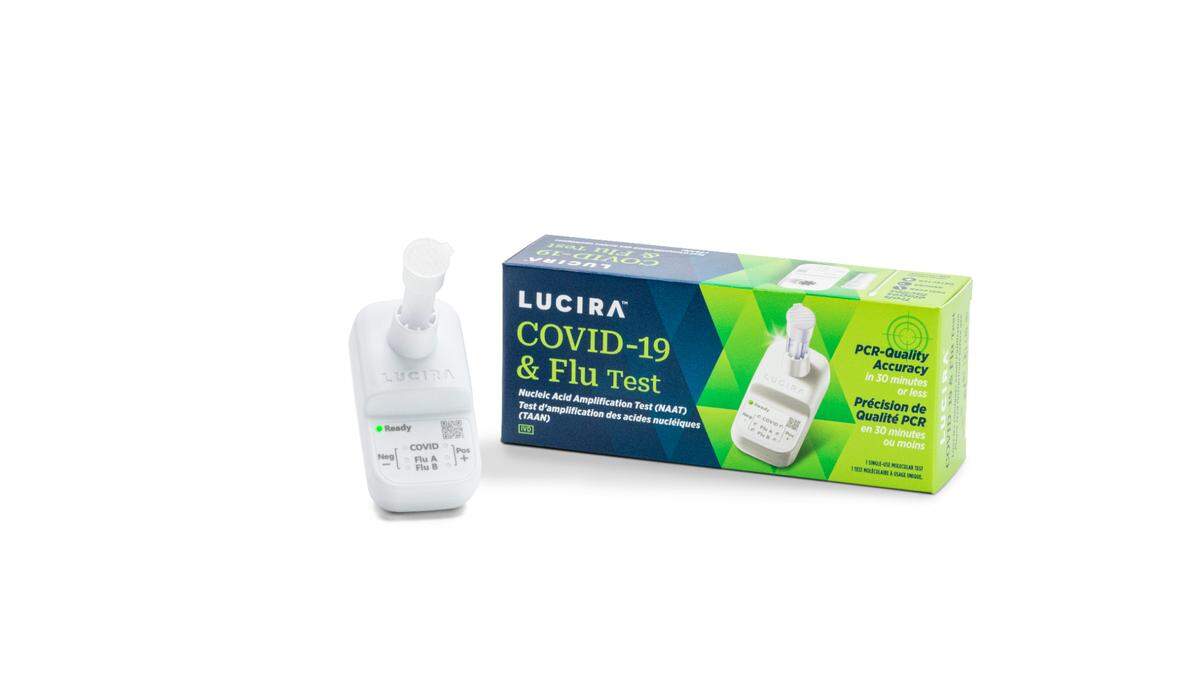
The Food and Drug Administration announced Friday it gave “an emergency use authorization” to an over-the-counter at-home test for both COVID and the flu from the company Lucira Health.
It is “the first over-the-counter at-home diagnostic test that can differentiate and detect influenza A and B, commonly known as the flu, and SARS-CoV-2, the virus that causes COVID-19,” the agency said in a statement announcing the approval.
The test is “a major milestone in bringing greater consumer access to diagnostic tests that can be performed entirely at home,” Jeff Shuren, director of the FDA’s Center for Devices and Radiological Health, said.
It “provides results from self-collected nasal swab samples in roughly 30 minutes,” according to the FDA.
Users swab their own noses and then swirl the swab “in a vial that is placed in the test unit.”
“The test unit will display the results that show whether a person is positive or negative for each of the following: Influenza A, Influenza B and COVID-19,” the FDA says.
The at-home test provides “immediate testing and accurate PCR (polymerase chain reaction) test results for all three viruses,” Lucira Health said in a statement.
Lucira Health announced in November the FDA had approved its combination COVID and flu test for “for point-of-care use,” such as in a doctor’s office.
When will the at-home flu and COVID test be available?
Lucira Health will soon announce when its new at-home test will be on the market in the U.S., the company’s CEO told USA Today.
The dual test is already available in Canada.
How much will the combo flu and COVID test cost?
Lucira Health has also not yet announced how much its dual test will cost in the U.S.
In Canada, a single-use test costs $98 in Canadian dollars. That’s about $72 per test in U.S. dollars.
Where will the test be available in Charlotte?
The new at-home flu and COVID test is not yet for sale in the U.S., but COVID tests from the same company are already available through major retailers such as Amazon and CVS.
Some Charlotte-area clinics and pharmacies do already offer a combined flu and COVID test, but you’ll need to make an appointment.
How reliable are at-home COVID tests?
Initial results show the new dual test is effective at detecting both COVID and the flu.
“In individuals with symptoms, the Lucira COVID-19 & Flu Home Test correctly identified 99.3% of negative and 90.1% of positive Influenza A samples, 100% of negative and 88.3% of positive COVID-19 samples and 99.9% of negative Influenza B samples,” the FDA said.
“Since there are currently not enough cases of Influenza B circulating to include in a clinical study, validation confirmed that the test can identify the virus in contrived specimens, and the EUA requires Lucira to continue to collect samples to study the test’s ability to detect Influenza B in real-world settings,” the agency added.
Still, inaccurate results can happen, the FDA notes.
“As with all rapid diagnostic tests, there is a risk of false positive and false negative results,” the agency says. “Individuals who test positive for either flu or COVID-19 should take appropriate precautions to avoid spreading the virus and should seek follow-up care with their physician or healthcare provider as additional testing may be necessary.”
This story was originally published February 27, 2023 at 4:42 PM.