A company says a counterfeit version of its COVID home test kit is on the U.S. market
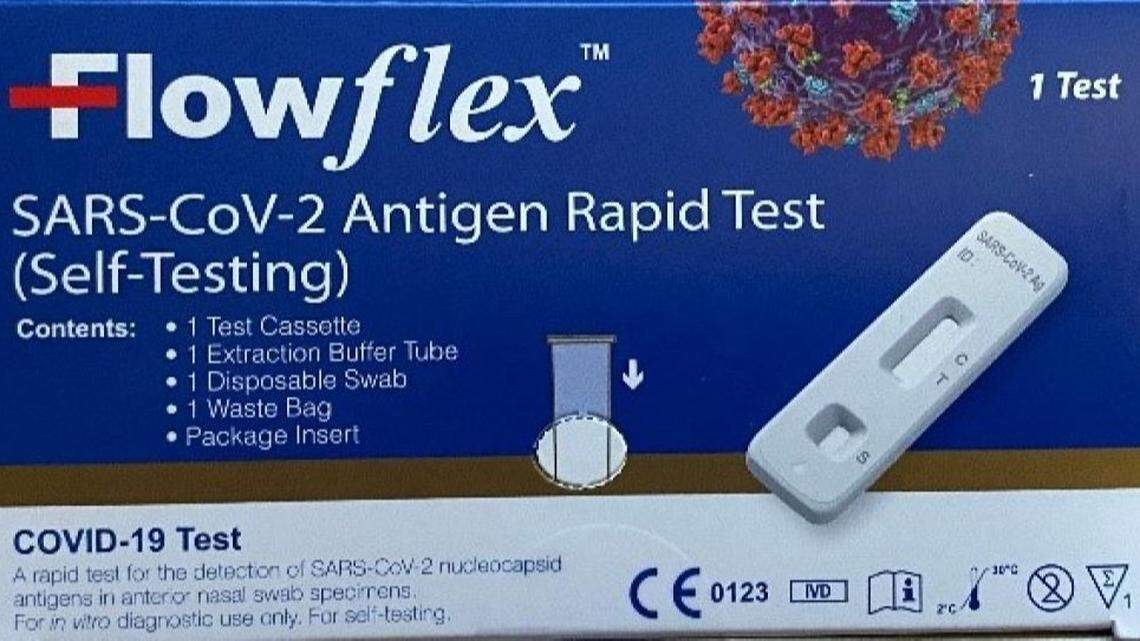
ACON Laboratories wants U.S. consumers to use its “Flowflex COVID-19 Antigen Home Test,” not its “Flowflex SARS-CoV-2 Antigen Rapid Test (Self-Testing)“ because the latter is a counterfeit test — in the United States.
As ACON explained in its FDA-posted recall notice, it knows about “the U.S. distribution of unauthorized, adulterated and misbranded counterfeit product” with the Flowflex SARS-CoV-2 packaging.
“ACON Laboratories is not importing the “Flowflex SARS-CoV-2 Antigen Rapid Test (Self-Testing)” into the U.S. as it is only authorized for sale in Europe and other markets,” the alert says.
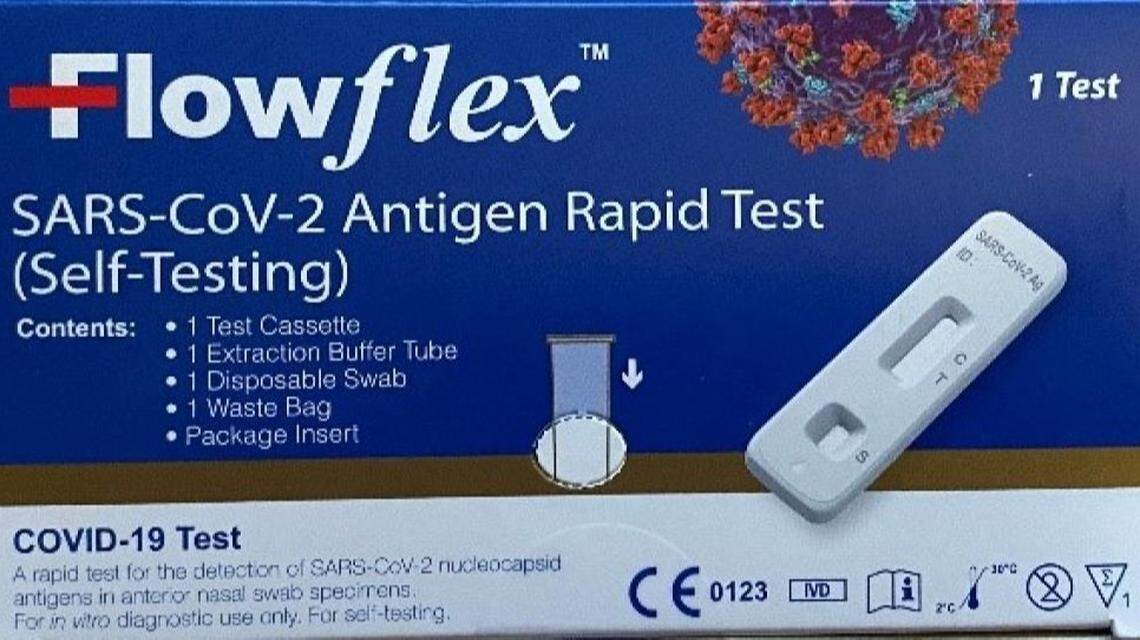

“If you have received the “Flowflex SARS-CoV-2 Antigen Rapid Test (Self-Testing)” with the blue box in the U.S. market, you should stop using this product and dispose of it,” ACON says. “The product has not been approved, cleared, or authorized for use in the U.S.”
Also, ACON would appreciate if you’d contact them at 800-838-9502 or flowflex_support@aconlabs.com and let them know where you got the counterfeit test.
Also, the FDA would like to know via its MedWatch Adverse Event page or by filling out a form you can get by calling 800-332-1088.
This story was originally published March 13, 2022 at 12:22 PM with the headline "A company says a counterfeit version of its COVID home test kit is on the U.S. market."
